|
Gas phase synthesis of transition metal oxides |

|
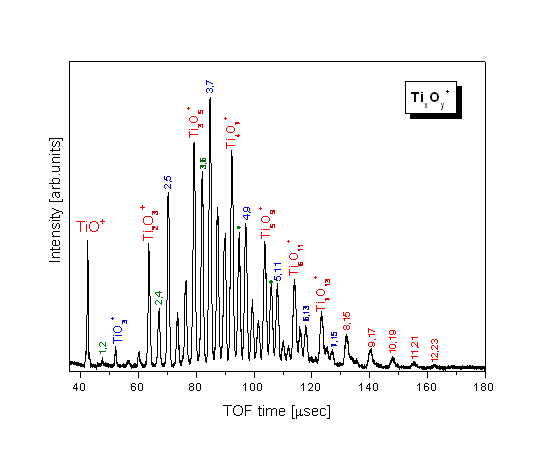
TOF-Mass Spectrum of TixOy+ Clusters 3 groups of stable TixOy+ clusters: (TiO2)n+ (TiO)+(TiO2)n (TiO)+(TiO2)nO2
Photofragmentation spectra (@ 308 nm) —> explain the growth of the clusters from TiO+ and Ti2O3+ by sequential uptake of TiO2 building blocks |
|
Titanium oxide clusters - Photodissociation |
|
Non-resonant photofragmentation is not very efficient Photons are replaced with atoms => lead to fragmentation through collision |
|
References:
M. Velegrakis et al Applied Physics A 97 (2009) 765
Maria Jadraque, Borja Sierra, Antonis Sfounis, and Michalis Velegrakis, “Photofragmentation of mass-selected titanium oxide cluster cations”, Applied Physics B, 100 (2010) 587
|

|
· Important species with potential applications in the fields of: - Physics (magnetism, nanoelectronics, ...) - Chemistry (catalysis, ...) - Biology (antibacterial properties, ...) · Their properties strongly depend on the local environment of the metal atoms such as the coordination number and the oxidation number. · These change as the cluster size increases. |
|
Transition metal oxide clusters-Examples
|


|
Ta: [Xe]5d36s2 ; Ta+: 5d36s1 Oxidation numbers Ta:+5,+4,+3,+2,+1 Ta+:+4,+3,+2,+1
|
|
Oxidation number Oxygen: - 2 Metal: + σ
|
|
Main Series: [Ta+4O-22]+ · [Ta+52O-25]n , [Ta+42O-24] +· [Ta+52O-25]n
|
|
Zr:[Kr] 4d25s2 Oxidation numbers Zr: +4,+3,+2,+1 Zr+: +3,+2,+1
|
|
Main Series: [Zr+2O-2]+ · [Zr+4O-22]n
|
|
Stability Rule: Neutral cluster : x · σ = y· 2 Cation(+1) : x · σ - 1 = y· 2 Anion (-1) : x · σ + 1 = y· 2
|